#AboutUs
- Home
- AboutUs
#Profile
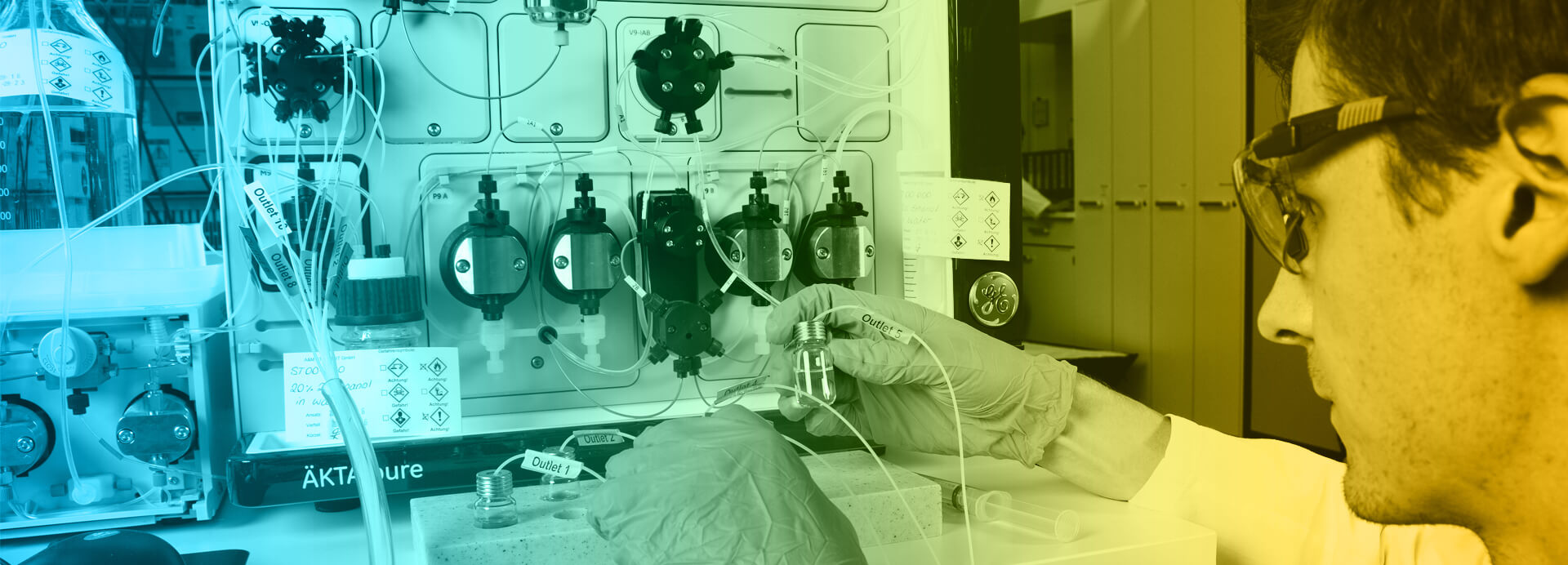
A&M STABTEST Labor für Analytik und Stabilitätsprüfung GmbH was established in 1995 by Dr Donald N. Dill, Dr Hans J. Diehl, Dr Hartmut Fischer and Dr Axel Römer as a spin-off from A&M Labor für Analytik und Metabolismusforschung GmbH. All founders are scientists with decades of experience in drug development. It is thanks to them, a corporate culture was installed, in which result-oriented work for our customers and quality are the top priorities. This approach has made us one of the largest GMP service providers for pharmaceutical analytics in Europe in recent decades. Since our foundation, we have been constantly adapting our GMP system to current regulatory requirements. The success of these efforts is regularly confirmed by GMP certifications and, since 2001, by inspections conducted by the US-FDA.
Our pronounced sense of quality and integrity is not only appreciated by regulatory authorities, but is also shared with our clients. This is why we have been providing or services to many of them since our founding years.
Working in continuous dialogue with our customers, we develop and expand our services in order to continue to be perceived as a competent partner in the innovative environment of pharmaceutical research. Within our organisation we work in specialized groups; for the storage and logistics of test items, the analysis of small molecules or proteins, mass spectrometry, the performance of bioassays or analytical development. All in order to satisfy the high quality requirements of our clients and international regulatory authorities.
We are proud, that we are currently Central Europe’s largest provider in the field of pharmaceutical analysis. And to ensure, that this remains the case, we not only continuously invest in modern analytical equipment and technologies, and in the continuous training and education of our employees, we also maintain a constant dialogue with our customers regarding future innovations in pharmaceutical research and development. For example, over 20 years ago we successfully supported the transformation of the pharmaceutical industry from chemically synthesised to biotechnologically produced active ingredients, and together with our innovative partners we are now developing analytical solutions for the next generation of cell- and gene-therapies.
#Values
Providing patients with efficacious and safe medicines, drives us to give our very best, every day. We achieve this not only with state-of-the-art lab equipment, but above all, with a well-trained team, in which all individuals take responsibility for themselves and their team. In order for our customers to acknowledge us as their extended workbench, each individual is obligated to carefully and comprehensibly document their efforts, and to proactively offer solutions to problems on par with our clients.
We establish trust through transparency and create genuine added value through our experience, expertise and personal commitment. Personalities with individual strengths and weaknesses form a group. A good team, however can only develop and thrive in the right environment. An environment in which, weaknesses are respected and strengths are promoted, in order to pursue our common objective – to over the best service in industry. We establish this environment by enabling a secure workplace, an interesting and meaningful occupation, further training and development opportunities for all, a family friendly workspace, as well as fair and trusting teamwork. This we do every day!
Do you share our values and believe you are an asset for the A&M team? Then click here to go to the careers area!
#Management
In 1995, four chemists laid the foundations for our past and present success story. To transport the success into the next decades to come. The torch has been passed on in 2020 to the next generation of long-standing and committed A&M colleagues. Supported by a strong and dedicated team, we will keep up the good work!
Dr. Hans J. Diehl
Managing Director
Quality
Dr. Donald Dill
Managing Director
Finances
Björn Kümer
Managing Director
Operational Business Mainz
Dr. Lejon Martens
Managing Director
Operational Business Bergheim
Dr. Steven Watt
Managing Director
Business Development Bergheim / Mainz
#Sites
A&M STABTEST GmbH
Kopernikusstr. 6
50126 Bergheim
+49 (0) 2271 7559-100
A&M STABTEST GmbH
Galileo-Galilei-Straße. 28
55129 Mainz
+49 (0) 6131 95978-0